More deficiencies uncovered at blood plasma donor centres in Canada

By Marty Logan
March 27, 2026
Note: I updated this post Friday night to fix the dates of the deaths in 2025 and 2026.
After two deaths in Winnipeg in the last six months following plasma donations, Health Canada has confirmed that a third death happened in the city on June 1, 2018 at a donor centre on Innovation Drive owned by Prometic Plasma Resources. The facility was bought from the company Kedrion by Spanish healthcare giant Grifols in 2022.
Health Canada received the report of the death on June 4, 2018. Its “assessment of the report has not identified a link between the death and the plasma donation. We cannot comment further,” Health Canada Spokesperson Mark Johnson told me in an email.
One of two recent plasma-related deaths in Winnipeg occurred following a donation at the Innovation Drive centre on Jan 30, 2026; the second took place after a donation at the Taylor Avenue location on Oct 25, 2025. Both centres are owned by Grifols, which runs 17 donation facilities in Canada.
Health Canada has not released the name of the first donor who died. The second death was that of university student Rodiyat Alabede.
Criticism is growing over Grifols’ for-profit donor operations after the recent deaths and a series of critical inspection reports by Health Canada.
A donor centre in Regina received a “non-compliant” assessment in January following a similar finding at a centre in Calgary in December. A third inspection is ongoing in an Oakville centre, but preliminary results note “deficiencies”.
Manitoba health minister calls for transparency
In a statement to the Globe and Mail on Thursday, Manitoba Health Minister Uzoma Asagwara said, “We expect a thorough and transparent investigation, including a clear understanding of whether all safety protocols were followed.
“All actions remain on the table,” the minister said about a possible ban on paid plasma donations in Manitoba. “People who donate plasma need to know their safety will never be put at risk.”
On Thursday the House of Commons committee on health grilled Grifols vice-president of sales in Canada, Mary Hughes, about the deaths and reports that the company could be violating its agreement with Blood Services Canada by selling plasma products outside the country.
“I’ll repeat, and to be clear: Today, there is no correlation between death and plasma collection,” Ms. Hughes replied to a question, reported The Globe.
After the Manitoba deaths were revealed, Grifols told CBC that an internal review was conducted and “based on the information available at this time, we have no reason to believe that there is a correlation between the donors’ passing and plasma donation.”
On Friday 20 March, CBC reported that the donation centre in Regina was inspected by Health Canada and found to be ‘non-compliant’ because, in part, it allowed a donor to give plasma twice in less than 48 hours.
A screenshot from the Health Canada January 2026 inspection report of the Grifols donor centre in Regina.
In a statement Grifols told me:
“Health Canada’s robust inspection framework and associated regulations help us to ensure that we are operating within their guidance and in pursuit of continuous improvement. This includes clarifying the waiting period between donations from two days to 48 hours.”
That’s when I started digging around, and found that the list of non-compliant items in the Regina report is longer than I imagined after reading the CBC article. And that there are other non-compliant Grifols plasma donation centres:
- The centre in Calgary was found non-compliant in an inspection that started on Dec 2, 2025 and ended on Dec 9.
- The centre in Oakville (the site of Grifols Canadian headquarters) has what the inspection reports call deficiencies, according to preliminary results of an inspection that started on Jan 28, 2026.
According to Health Canada, none of the observations at the Regina and Calgary centres were considered critical. “Based on the information that Health Canada has received and reviewed, the integrity of the blood components that were drawn by the Grifols’ sites in Calgary and Regina are not compromised, and the two sites were permitted to remain open,” said the email.
Health Canada reviewing corrective action plans
Health Canada added that following the non-compliant reports in Regina and Calgary it requested action plans from Grifols. “Health Canada is currently reviewing the corrective action plans and will request additional information, if needed.”
The Health Canada email added: “Not all inspections rated non-compliant are because of a critical observation. For example, non-compliant ratings can also be the result of non-critical observations, repeat observations, and/or when an establishment is not in control of its regulated activities.
Plasma is the pale-yellow liquid component of blood, which makes up about 55% of the total volume of a person’s blood. Plasma donation consists of collecting a donor’s blood, separating the plasma portion from the blood cells, and then returning the blood cells to the donor. It takes 1-2 hours.
Grifols opened its first donor centre in Winnipeg in 2022. Also that year, it announced a 15-year agreement with Blood Services Canada, the national blood authority, to collect plasma donations.
In 2020, Grifols bought existing Montreal plasma-processing plants for more than $500 million. The company says they should be fully operational this year.
According to its website, Grifols pays honorariums of up to $100 for donations plus occasional bonuses that can reach $100. In contrast, donating blood in Canada is voluntary; donors are not paid.
The company says that after developing corrective action plans for the Calgary and Regina centres it “began implementation immediately with a focus on preventing recurrence and strengthening overall compliance.”
The two centres are operating while they take action, adds Grifols.
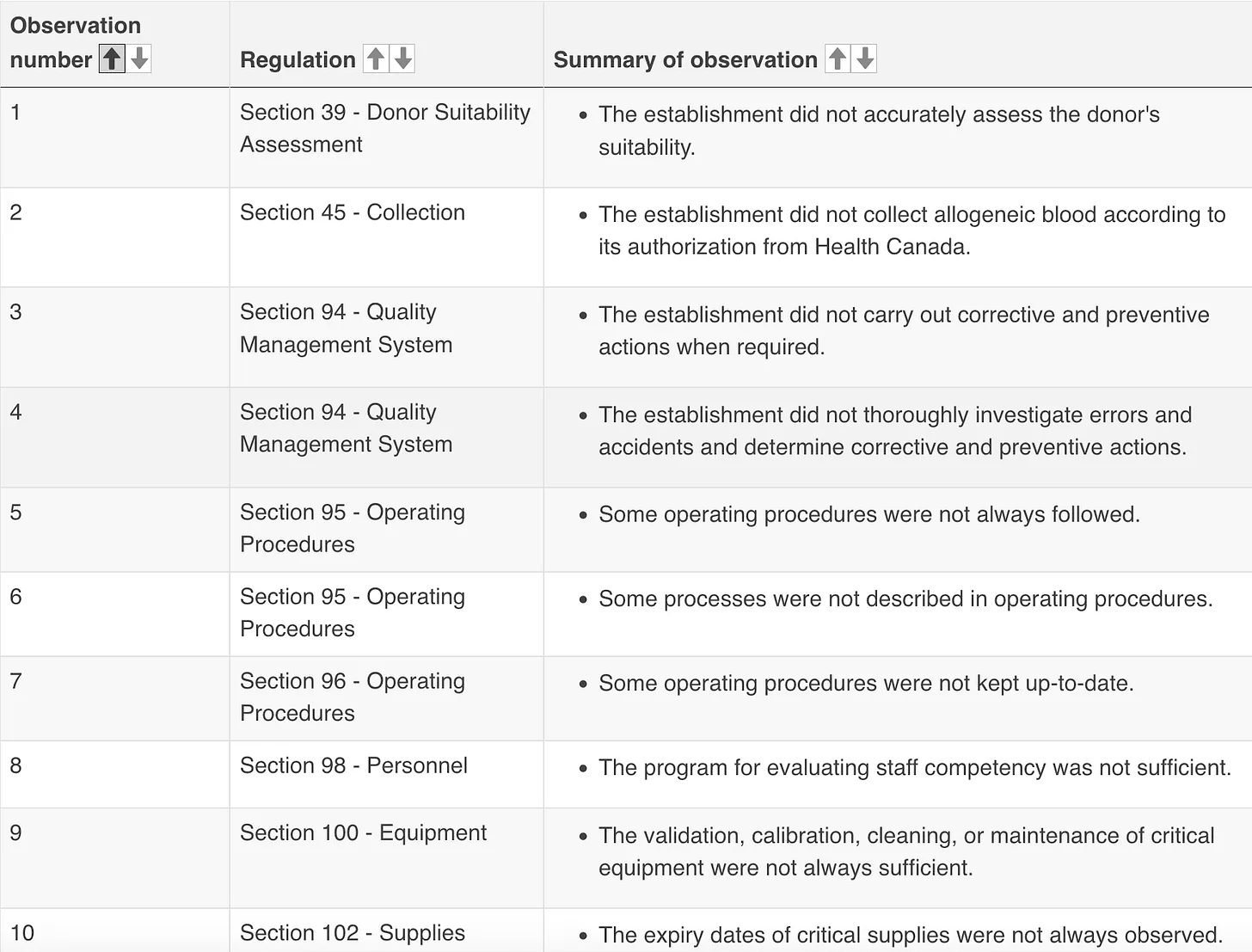
In addition to the deficiencies that Health Canada inspectors found in Grifols’ Calgary, Regina, and Oakville centres, inspections in Saskatoon (2023) and Saint John (2022) also found the centres non-compliant. All five reports have deficiencies in common including:
- The centres did not thoroughly follow up errors and accidents to prevent future repeats or complete required reporting of incidents;
- Operating procedures were not always followed, some were not kept up-to-date, and the centre in Saskatoon “did not have operating procedures for all of the activities it conducts related to human safety and the safety of blood.”
Common issues reported in the latest inspections, at the Oakville, Regina, and Calgary centres, include:
- Insufficient “donor suitability assessment”;
- Concerning personnel— deficiencies in personnel qualifications or training, staff evaluations, and programmes for staff orientation or training.
